Proposal (875) to South American Classification Committee
Elevate Penelope
bridgesi to species status and endorse an English name
Effect
on SACC List:
A positive vote would add Penelope bridgesi to the SACC list.
Background. The only SACC note on
either taxon, bridgesi or bronzina, which represent the only
recognized subspecies of Dusky-legged Guan P. obscura, is as follows:
14a. Evangelista-Vargas
& Silveira (2018) treated the subspecies bridgesi of Bolivia and Argentina as a separate species but based
on PSC rationale.
Penelope bridgesi
was described as a full species by Gray (1860) with the type locality of
“Bolivia”, collected by Thomas C. Bridges; holotype in the British Museum (NHM
1846.9.9.42). It was subsequently lumped with P. obscura by
Ogilvie-Grant (1893) upon his specimen examination involving seven specimens of
obscura and the bridgesi holotype. This arrangement has remained
the status quo ever since, although often questioned e.g. Hartert &
Venturi 1909, who described bridgesi as being “bien distincte”, besides
many other ornithologists describing numerous plumage distinctions and the fact
that bridgesi is notably larger than obscura (Delacour &
Amadon 2004). Note also that bridgesi lives primarily in Yungas forest
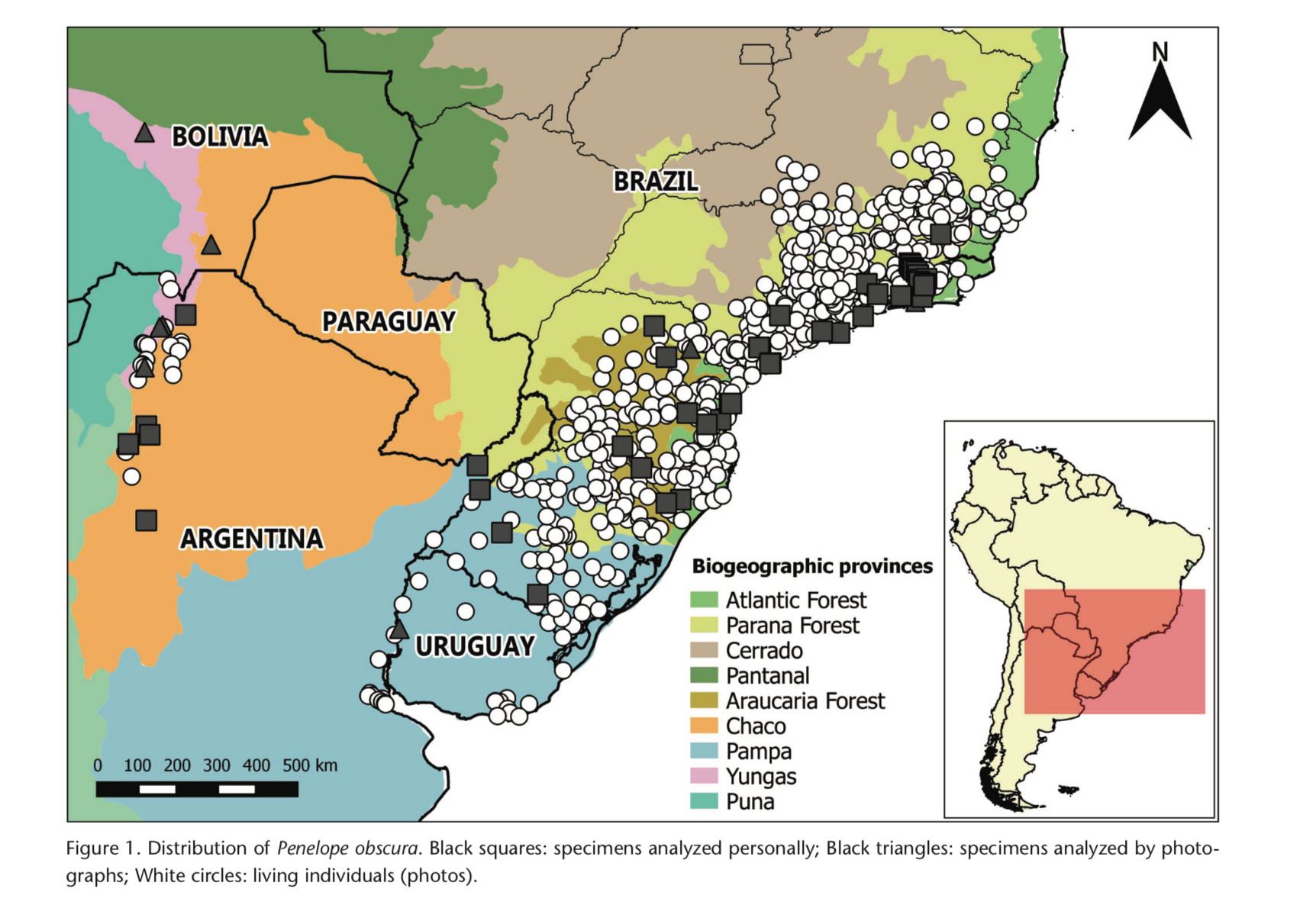
and ranges exclusively from southern Bolivia through north-west Argentina.
In contrast bronzina was
described as a subspecies of P. obscura by Hellmayr (1914) from Colônia
Hansa, Santa Catarina, Brazil, and the two taxa are parapatric in south-east
South America and possibly grade into one another. The status of bronzina
as a subspecies has largely gone unquestioned except that some authors suggest
a merger with obscura (see New
Information).
Penelope obscura
(together with bronzina) is
allopatric by ca.1600 km from bridgesi, and the two live in very different forest types; obscura in
gallery forest from the Parana Delta northwards and bronzina also in
gallery forest as well as patches of Araucaria forest, in strong contrast to bridgesi, which lives in subtropical Yungas forest in the Andean foothills. The
notion that bridgesi also lives in the chaco (Evangelista-Vargas
& Silveira 2018) is in error and refers to the foothill intergrade between
sierran chaco and Yungas forest, yet this habitat has nothing to do with the
dry or humid chaco ecotone.
It seems extraordinary that
two so-very-different looking species have been lumped for well over a century
with so little discord.
New Information. A study of 104
specimens of bridgesi, obscura and bronzina
(Evangelista-Vargas & Silveira 2018) confirmed what was already known on
their strong allopatry, and biometric tests revealed no overlap in wing
measurements between obscura (including bronzina) and bridgesi,
with bridgesi being longer.
Their
plumage analysis noted that “all specimens attributed to P. o. bridgesi lacked
olive brown coloration with a green tinge to the upperparts (unlike P. o.
obscura and P. o. bronzina). Instead, specimens of P. o. bridgesi
showed chestnut upperparts contrasting with the black head. The underparts
were consistently uniform and slightly paler than the back, whereas in P.
o. obscura and P. o. bronzina the coloration of the upperparts was
highly variable. The wing coverts of P. o. bridgesi show broad white
margins, a character state not found in P. o. obscura and P. o.
bronzina, which have indistinct white margins to the wing coverts.” Such
statements reiterate the type description that bridgesi has different
contour plumage coloration, a very different distribution and intensity (width)
of white streaking on different parts of its body; in particular the
wing-coverts and breast. This can be clearly seen in these pictures of bridgesi (https://macaulaylibrary.org/asset/31599331, https://macaulaylibrary.org/asset/199545731), and obscura/bronzina (https://macaulaylibrary.org/asset/133418341,https://macaulaylibrary.org/asset/139191981), and is also
appreciable in specimens as shown in Figure 5 of Evangelista-Vargas
& Silveira (2018).

Evangelista-Vargas
& Silveira (2018) also provided re-descriptions of obscura and bridgesi
as follows:
Penelope obscura Temminck, 1815
“Re-description. No sexual dimorphism
found in plumage coloration. Whitish edges extension of head feathers varies
from wide to almost imperceptible. Some individuals show a moderately distinct
superciliary stripe formed by short feathers with white edges. Overall color of
crown feathers variable, ranging from black, gray to brown. White hue of the
edge of the feathers varies individually. Brown back may show greenish,
brownish or blackish tinge. Neck, breast, upper back, and shoulders conspicuously
edged with white. Birds in the northern parts of the species’ range tend to
have more noticeable white streaks on the back, but streaks also may be
lacking. The coloration of the underparts is variable and may show a
contrastingly darker chest than belly, but underparts may also be uniform dark
or lighter than the breast. The belly feathers have contrasting horizontal
scaling, which may range from brown to orangish, and may vary in intensity.
Tail feathers are dark brown with bronze or purplish sheen. Bare skin of face
slaty, legs dusky gray to blackish, and wattle red.”
Penelope bridgesi Gray, 1860
“Re-description. No sexual dimorphism
found in plumage coloration. General coloration chestnut, with limited individual
variation. Crest feathers dark with whitish margins, most strongly marked on
the forehead; little or no trace of an superciliary stripe. Upperparts
chestnut, contrasting with the black head and hindneck. Lower parts uniform
dull reddish brown and slightly paler than the back. The belly feathers have
contrasting horizontal scaling, which may range from brown to orangish and may
vary in intensity. Feathers of the neck, mantle and chest are margined with
white, wider on the mantle and wing coverts. Tail feathers dark brown with
brownish sheen. Bare skin of face slaty, legs dusky gray to blackish, and
wattle red.
Furthermore,
Pearman (in del Hoyo and Motis (2004)) called attention to vocal distinctions:
“The excited calls or alarms of bridgesi
(Salta Prov.) are low-pitched whistles mixed with guttural rasps, e.g., “SWig
SWI-u, SWig-u KREG, swig-u, u, KREG KREG KREG” etc.; the loud, guttural “KREG” notes are given with
more regularity when alarmed, cf. (op. cit.), the calls or
alarm of the nominate race [i.e. obscura] (NE Buenos Aires Prov.) are
higher pitched, disyllabic and lack the guttural notes of bridgesi, sounding more like: “Wikik Wikik-u, Wikik-u-u, Wikik,
Wikik-u” etc.
Discussion
In
our own extensive personal experience of obscura and bridgesi, it
seems incredulous that they are still regarded as a single species, on the
basis of a single unscrupulous museum comparison more than a century ago. We
also concur with others that bridgesi is a very much larger guan than obscura
as borne out by total length measurements taken at the time of collection (MACN
and IADIZA specimen data):
obscura: 480-660 (n=2)
bridgesi: 770-890 (n=9)
Recommendation. We recommend a YES
vote to elevate P. bridgesi to species status.
English
name.
Thus far, only the name Bridges’s Guan has been suggested as an English name
(Evangelista-Vargas & Silveira 2018) in reference to the collector of the
holotype. Please note the implications
of pronunciation since Bridges is a plural surname. As an alternative we
promote the name Yungas Guan because bridgesi is the abundant species of
guan within its range in Yungas forest. The only other guan species in range is
the very localized Red-faced Guan P. dabbenei, which occurs at higher
elevations in the alder treeline zone. As such, the common guan species of the
Yungas forest is most certainly bridgesi.
We
recommend a YES vote to endorse the name Yungas Guan. A NO vote would endorse
Bridges’/Bridges’s Guan.
References
Delacour, J. & Amadon, D. (2004) Curassows and
Related Birds. Lynx Edicions, Barcelona/The National Museum of Natural
History, New York.
del Hoyo, J. & Motis, A. (2004) Update Chapter. Pp. 322-476
in: Delacour, J & Amadon, J. 2004 Curassows and Related Birds. Second
Edition. Lynx Edicions and NMNH, Barcelona & New York.
Evangelista-Vargas
& Silveira,
L. F. (2018) Morphological evidence for the taxonomic status of the Bridges’s
Guan, Penelope bridgesi, with comments on the validity of P. obscura
bronzina (Aves: Cracidae). Zoologia 35: e12993.
Gray, G.R. (1860)
Synopsis of the species of the genus Penelope. Proc. Zool. Soc. London
28: 269-272.
Hartert E. &
Venturi S. (1909) Notes sur les oiseaux de la république Argentine. In:
Rothschild W, Hartert E, Jordan K (Eds) Novitates Zoologicae XVI. Zoological
Museum of Tring, London and Aylesbury, 159–267.
Hellmayr
1914 Critical Notes on the types of
little-known species of Neotropical birds. Part 3. Novit. Zool. 21: 158-179.
Ogilvie-Grant W.R.
(1893) Catalogue of the Birds in the British Museum 22. Order of the
Trustees, London.
M. Pearman and J. I. Areta, August 2020
Note
from Remsen on voting:
Let’s treat this as two separate subproposals, with “A” being species limits
and “B” being English name if A passes.
Comments from Remsen:
“A. YES. On the one hand, size and plumage in general
are not characters that are sufficient to define species. Disjunct distribution is also not a
sufficient criterion for species range; the degree of “disjunctness”
varies on a continuous scale that has no defensible threshold for species rank. In fact, the size differences would be
predicted within populations of the same species on the basis of Bergmannian patterns:
the higher elevation taxon would be predicted to be larger, as is the
case here. Also, habitat differences per
se are essentially useless as sufficient criteria for species limits given the
notorious flexibility of habitat preferences within many monotypic species. The vocal differences are potentially
sufficient criteria on their own, but I would be cautious in using them without
a formal study of the range of variation in each species, with N etc. Nonetheless, I vote in favor of the
split. Although I would have to check specimens
to be sure, it seems clear from the descriptions that the plumage differences
between these two are greater than between some taxa ranked as species in Penelope
(e.g., P. argyrotis and P. barbata), in which the degree of
interspecific plumage variation in general is unimpressive. As pointed out in the proposal, their
maintenance as conspecifics seems largely a case of historical momentum with
virtually no critical analysis. Delacour
& Amadon of course mention the differences between the taxa but made no
remarks on whether they might be separate species; however, both authors were
notorious lumpers. At this point, the
proposal and the Evangelista-Vargas & Silveira paper has me convinced that
the burden-of-proof falls on conspecificity.
In fact, whether they are sister taxa should also be investigated
genetically; I assume from Evangelista-Vargas & Silveira that no phylogeny
has included samples from both taxa.
“B. YES on Yungas
Guan. In addition to the great
differences in range size, I also consider it unresolved as to whether they are
sister taxa, so I have even less concern than usual for the “parent-daughter”
guidelines of English names.”
Comments
from Stiles:
“YES to A: morphology, vocalizations, ecology and distribution justify the
recognition of bridgesi as a species;
YES to B: Yungas Guan is short and appropriate.”
Comments
from Claramunt:
“YES. In the context of the very conservative Penelope,
those few differences are remarkable.”
Comments
from Robbins:
“ A) YES, to recognizing bridgesi as a species given that plumage
morphology differentiation is greater between it and obscura than in
other presumed Penelope sister taxa that are recognized as species. The
vocal differences described by Pearman are also suggestive for species
recognition.
Comments from Zimmer: “(A) YES.
With a nod to each of the caveats mentioned by Van, I still think the
plumage differences between bridgesi
and obscura/bronzina meet the
“yardstick” provided between other species pairs with Penelope, particularly when combined with the described vocal and
morphometric differences, habitat specialization, and range disjunction. (B) YES to “Yungas Guan” for bridgesi, and retain “Dusky-legged Guan”
for obscura + bronzina.”
Comments from Bonaccorso: “ A – YES. Given the
evidence gathered, this is repairing a historical mistake, and this is not an
exaggeration.”
Comments from Jaramillo: “A. YES – These birds are quite different for Penelope,
the size difference alone is perhaps an impediment to interbreeding would they
ever meet in life. B. YES – Yungas Guan is the better name.”